TB-500 is one of the most discussed peptides in injury recovery and athletic performance circles.
It’s frequently mentioned alongside BPC-157 as a potential tool for tissue repair. But while they’re often grouped together, TB-500 works through different biological pathways and carries its own risks and unknowns.
If you’ve seen claims that TB-500 can accelerate healing, reduce inflammation, or support muscle and tendon repair, you’re not alone.
The bigger question is: what does the research actually show?
This guide covers what TB-500 is, how it’s thought to work, what evidence exists, potential side effects, dosing considerations, and its legal status in 2026.
What Is TB-500?
TB-500 is a synthetic version of Thymosin Beta-4 (Tβ4) — a naturally occurring peptide found in many human and animal cells.
Thymosin Beta-4 plays a role in:
- Tissue repair
- Wound healing
- Cell migration
- Blood vessel formation
TB-500 is the lab-created version used in research settings. It’s designed to be more stable and easier to administer outside the body.
Important distinction:
TB-500 and Thymosin Beta-4 are often used interchangeably, but technically TB-500 refers to the synthetic research compound, while Tβ4 is the naturally occurring peptide.
TB-500 is not FDA-approved for human use.
How Does TB-500 Work?
Most proposed mechanisms are based on research into Thymosin Beta-4.
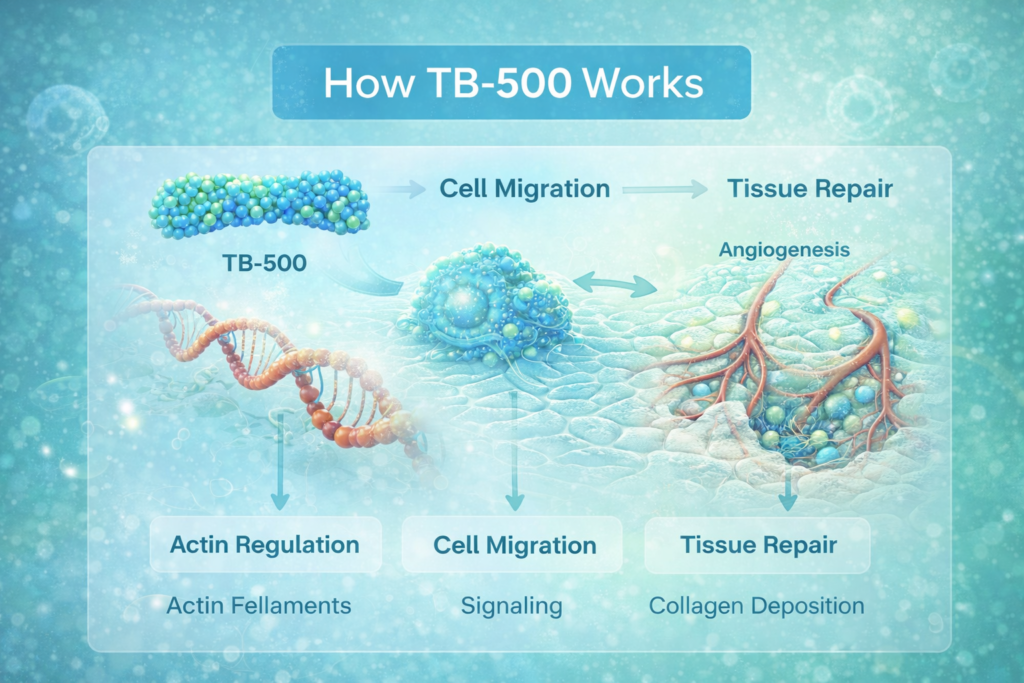
Potential mechanisms include:
Cell Migration (Chemotaxis)
TB-500 may promote the migration of cells such as endothelial cells and keratinocytes to injured areas. This process is essential for wound closure and tissue repair.
Actin Regulation
TB-500 binds to actin, a structural protein that plays a key role in cell movement and shape. By influencing actin dynamics, TB-500 may affect how cells respond to injury.
Anti-Inflammatory Effects
Some studies suggest Tβ4 has anti-inflammatory properties, which could support healing by limiting excessive inflammatory response.
Angiogenesis Support
Like BPC-157, TB-500 may promote angiogenesis — the formation of new blood vessels — which is critical for delivering oxygen and nutrients to damaged tissue.
Collagen and Tissue Remodeling
Preclinical research suggests it may influence collagen deposition and reduce fibrosis (scar tissue formation).
Most of this evidence comes from animal studies and laboratory research. Human clinical data remains extremely limited.
What Do People Use TB-500 For?
Despite the lack of large human trials, TB-500 is discussed for:
- Muscle strains and tears
- Tendon and ligament injuries
- Chronic overuse injuries
- Post-surgical recovery
- Joint pain
- General recovery between training sessions
Some anecdotal reports also mention hair growth, though this area is not well studied in humans.
It’s important to emphasize:
These uses are based largely on animal research and user reports — not established medical guidelines.
TB-500 vs. BPC-157
TB-500 is often compared to BPC-157, and some people use both together.
Here’s how they differ:
Mechanism
- BPC-157: Often associated with localized tissue healing and growth factor modulation
- TB-500: Associated more with systemic cell migration and actin regulation
Origin
- BPC-157: Derived from gastric protein fragments
- TB-500: Synthetic version of Thymosin Beta-4
Typical Emphasis
- BPC-157: Frequently discussed for gut health and targeted injuries
- TB-500: Often described as having broader systemic effects
Some users “stack” them, but there is no clinical evidence demonstrating that combining them produces superior outcomes.
For a broader comparison of performance-enhancing compounds, see our guide on peptides vs steroids.
What Does the Research Show?
TB-500 / Thymosin Beta-4 has been studied more extensively than many other research peptides, but most evidence remains preclinical.
Animal and laboratory studies suggest potential roles in:
- Wound healing acceleration
- Cardiac tissue repair after injury
- Corneal (eye) healing
- Muscle regeneration
- Reduced fibrosis
However:
- Human clinical trials are extremely limited
- Most research examines natural Tβ4, not TB-500 specifically
- Optimal dosing in humans is not established
- Long-term safety data does not exist
The honest assessment:
Preclinical data is promising. Whether those effects reliably translate to practical human use is still unclear.
How Is TB-500 Used?
TB-500 is most commonly administered via subcutaneous injection.
Reported usage patterns (based on anecdotal reports, not clinical standards) often involve:
- An initial “loading” phase over several weeks
- Followed by lower-frequency maintenance dosing
It is typically sold as a lyophilized powder that must be reconstituted before use.
Important:
There are no FDA-approved dosing guidelines. Information found online is based on user experimentation and extrapolation from animal research.
Anyone considering TB-500 should understand that use occurs outside formal regulatory oversight.
Potential Side Effects and Risks
Short-term anecdotal reports often describe TB-500 as well tolerated. That does not equal proven safety.
Reported side effects include:
- Fatigue
- Headaches
- Injection site irritation
- Mild nausea
- Flu-like symptoms (rare reports)
Theoretical Concerns
Cell proliferation risk
Because TB-500 promotes cell migration and angiogenesis, some researchers raise theoretical concerns about how it might interact with abnormal cell growth. This risk is speculative but worth acknowledging.
Long-term safety unknown
No long-term human trials exist.
Quality control concerns
As a research chemical, TB-500 is not subject to pharmaceutical-level oversight. Purity and dosing accuracy vary widely.
Anyone with a history of cancer or pre-cancerous conditions should approach with extreme caution.
Legal Status and Sports Regulations
TB-500 is not FDA-approved for human use.
In the United States:
- It can be sold as a research chemical
- It cannot legally be marketed for human consumption
- Possession for personal use is not explicitly criminalized
- There are no regulatory protections for users
For a broader breakdown, see our article on Are Peptides Legal in 2026?.
For Athletes
TB-500 is prohibited by the World Anti-Doping Agency (WADA) under the category of growth factors and related substances.
If you are subject to drug testing in any organized sport, you should assume TB-500 is banned.
Should You Try TB-500?
That depends on your risk tolerance and your situation.
You might consider it if:
- You have a chronic injury unresponsive to conventional approaches
- You understand the experimental nature
- You are not subject to drug testing
You should avoid it if:
- You want established, evidence-based treatment
- You have a history of cancer
- You are pregnant or nursing
- You are uncomfortable with unknown long-term risks
If considering TB-500, working with a qualified healthcare professional is strongly recommended.
Start conservatively. Avoid stacking multiple compounds initially. Monitor your response carefully.
The Bottom Line
TB-500 is one of the more extensively studied research peptides in the context of tissue repair — at least in animal models.
The preclinical evidence is intriguing. Human data is sparse. Long-term safety is unknown. Regulatory oversight is minimal.
It may represent a potential experimental option in certain situations, but it is not a proven therapy — and it is not without risk.
Approach it thoughtfully, with realistic expectations and a clear understanding of the uncertainties involved.

